Local Perspective
In Pakistan exact incidence of congenital hypothyroidism is not known as no routine newborn screening was performed until ZB Foundation. In a study performed in a tertiary care hospital of Karachi, incidence reported as 1:1000 (Khurram IA et al 2003).
In another study conducted retrospectively in Islamabad PIMS, high frequency of congenital hypothyroidism in hospital deliveries was reported. In three years of study, 962 babies out of 1337 babies were normal, 31 were borderline cases with values ranging from 10 to 18mU/ml and 3 babies were persistent positive cases. (Raza, H. et al 2013; Noreen, R. et al 2016)
Mumtaz Lakhani states in her article, Lakhani, M. U. M. T. A. Z., Khurshid, M. O. H. A. M. M. E. D., Naqvi, S. H., & Akber, M. O. H. A. M. M. A. D. (1989). Neonatal screening for congenital hypothyroidism in Pakistan. JPMA. The Journal of the Pakistan Medical Association, 39(11), 282-284.:
“The screening for congenital hypothyroidism was initiated in Pakistan by the Aga Khan University Hospital (AKUH) in March 1987. By April 1988, 5000 neonates were screened and five cases of congenital hypothyroidism were diagnosed. The study revealed the incidence of hypothyroidism to be one case per 1000 newborns which is about 4 times more than that in the West.”
Jabbar stated in Jabbar, A., Yawar, A., Waseem, S., Islam, N., Ul Haque, N., Zuberi, L., Khan, A., Akhter, J. (2008). Vitamin B12 deficiency common in primary hypothyroidism. Journal of the Pakistan Medical Association, 58(5), 258-61 :
“There is a high (approx 40%) prevalence of B12 deficiency in hypothyroid patients. Traditional symptoms are not a good guide to determining presence of B12 deficiency. Screening for vitamin B12 levels should be undertaken in all hypothyroid patients, irrespective of their thyroid antibody status. Replacement of B12 leads to improvement in symptoms, although a placebo effect cannot be excluded, as a number of patients without B12 deficiency also appeared to respond to B12, administration.”
Global Perspective
Congenital hypothyroidism affects an estimated 1 in 2,000 to 4,000 newborns. For reasons that remain unclear, congenital hypothyroidism affects more than twice as many females as males (Noreen, R., Memon, M. H., Murtaza, G., & Hanif, S. 2016).
In USA – The incidence of congenital hypothyroidism, as detected through newborn screening, is approximately 1 per 4000 births. An increase in the diagnosis of primary congenital hypothyroidism has been reported in New York. This trend has also been observed in some other states, although not all. Possible explanations include changing demographics of the birth population, including changes in race, ethnicity, and the incidence of low birth weight. Changes in laboratory and screening methodology may also play a role in this reported rise in incidence. Some infants identified as having primary congenital hypothyroidism may have transient disease and not permanent congenital hypothyroidism.
In Central Africa – where iodine deficiency occurs along with excess dietary Cyanate from cassava (Manihot esculenta) – as many as 10% of newborns may have both low cord blood T4 concentration and TSH concentrations over 100 mU/L.
Data from most countries with well-established newborn screening programs indicate an incidence of congenital hypothyroidism of about 1 per 3000-4000. Some of the highest incidences (1 in 1400 to 1 in 2000) have been reported from various locations in the Middle East.
Although percentages of specific etiologies vary from country to country, ranges are as follows:
- Ectopic thyroid – 25-50%
- Thyroid agenesis – 20-50%
- Dyshormonogenesis – 4-15%
- Hypothalamic-pituitary dysfunction – 10-15%
| Demographic | Incidence |
| Overall | 1:1681 |
| Gender | |
| Male | 1:1763 |
| Female | 1:1601 |
| Ethnicity | |
| White | 1:1815 |
| Black | 1:1902 |
| Asian | 1:1016 |
| Hispanic | 1:1559 |
| Birth weight | |
| < 1500 g | 1:1396 |
| 1500 – 2500 g | 1:851 |
| > 2500 g | 1:1843 |
| Single vs. multiple births | |
| Single | 1:1765 |
| Twin | 1:876 |
| Multiple | 1:575 |
| Mother’s age | |
| < 20 years | 1:1703 |
| 20-29 years | 1:1608 |
| 30-39 years | 1:1677 |
| > 39 years | 1:1328 |
Table 1: Demographic factors of CH and their incidences
What is Congenital Hypothyroidism?
Endemic Cretinism is Congenital Hypothyroidism?
The term endemic cretinism is used to describe clusters of infants with goiter and hypothyroidism in a defined geographic area. Such areas were discovered to be low in iodine, and the cause of endemic cretinism was determined to be iodine deficiency. In the 1920s, adequate dietary intake of iodine was found to prevent endemic goiter and cretinism. Congenital hypothyroidism is inadequate thyroid hormone production in newborn infants. It can occur because of an anatomic defect in the gland, an inborn error of thyroid metabolism, or iodine deficiency.
Is The Thyroid Gland The Only Effector?
Secondary congenital hypothyroidism can also occur as part of syndromes that affect other organs and tissues in the body. These forms of the condition are described as syndromic. Some common forms of syndromic hypothyroidism include Pendred syndrome, Bamforth-Lazarus syndrome, and brain-lung-thyroid syndrome.
Signs and Symptoms:
Infants with congenital hypothyroidism are usually born at term or after term. Symptoms and signs include the following:
- Decreased activity
- Large anterior fontanelle
- Poor feeding and weight gain
- Small stature or poor growth
- Jaundice
- constipation
- Hypotonia
- Hoarse cry
Often, affected infants are described as “good babies” because they rarely cry and they sleep most of the time.
The physical findings of hypothyroidism may or may not be present at birth. Signs include the following:
- Coarse facial features
- Macroglossia
- Large fontanelles
- Umbilical hernia
- Mottled, cool, and dry skin
- Developmental delay
- Pallor
- Myxedema
- Goiter
Anemia may occur, due to decreased oxygen carrying requirement. A small but significant number (3-7%) of infants with congenital hypothyroidism have other birth defects, mainly atrial and ventricular septal defects.
Pathophysiology
The thyroid gland develops from the buccopharyngeal cavity between 4 and 10 weeks’ gestation. The thyroid arises from the fourth branchial pouches and ultimately ends up as a bilobed organ in the neck. Errors in the formation or migration of thyroid tissue can result in thyroid aplasia, dysplasia, or ectopy. By 10-11 weeks’ gestation, the fetal thyroid is capable of producing thyroid hormone. By 18-20 weeks’ gestation, blood levels of T4 have reached term levels. The fetal pituitary-thyroid axis is believed to function independently of the maternal pituitary-thyroid axis.
Pathophysiology of CH
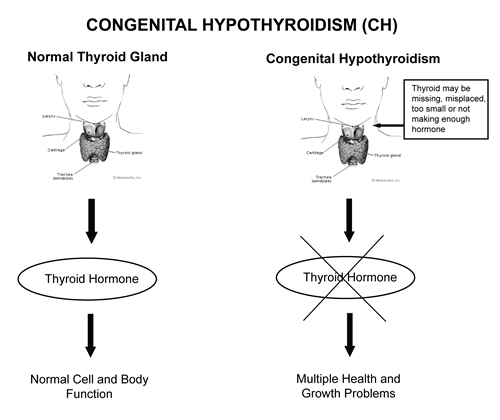
T4 is the primary thyronine produced by the thyroid gland. Only 10-40% of circulating T3 is released from the thyroid gland. The remainder is produced by monodeiodination of T4 in peripheral tissues. T3 is the primary mediator of the biologic effects of thyroid hormone and does so by interacting with a specific nuclear receptor. Receptor abnormalities can result in thyroid hormone resistance.
The importance of thyroid hormone to brain growth and development is demonstrated by comparing treated and untreated children with congenital hypothyroidism. Thyroid hormone is necessary for normal brain growth and myelination and for normal neuronal connections. The most critical period for the effect of thyroid hormone on brain development is the first few months of life.
Mortality/Morbidity
Affected infants whose treatment is delayed can have neurologic problems such as spasticity and gait abnormalities, dysarthria or mutism, and autistic behavior.
Race
Congenital hypothyroidism is observed in all populations. The prevalence at birth is increased in Hispanics, particularly in Hispanic females, who have a birth prevalence of 1 in 1886 births. Black infants have about one third the prevalence rate of white infants.
Sex
Most studies of congenital hypothyroidism suggest a female-to-male ratio of a 2:1. Devos et al showed that much of the discrepancy is accounted for by infants with thyroid ectopy. The sex ratio for Hispanics is more striking, with a 3:1 female-to-male ratio. The ratio is lower among Black infants.
Age
By definition, congenital hypothyroidism is present at, or before, birth. Children who develop primary hypothyroidism when aged 2 years or older have poor growth and slow mental condition but generally do not exhibit the profound and incompletely reversible neurologic abnormalities observed in untreated congenital hypothyroidism.
Inheritance Pattern
Most cases of congenital hypothyroidism are sporadic, which means they occur in people with no history of the disorder in their family. When inherited, the condition usually has an autosomal recessive inheritance pattern, which means both copies of the gene in each cell have mutations. Typically, the parents of an individual with an autosomal recessive condition each carry one copy of the mutated gene, but they do not show signs and symptoms of the condition.
Having CH
If they are healthy and developing normally, older brothers and sisters of a baby with CH are unlikely to have the condition. Talk to your doctor if you have questions about your other children.
Future Pregnancies
All 50 US states offer newborn screening for CH. However, when a previous child in the family has had CH, newborn screening results may not be sufficient to rule out the condition in a newborn baby. In this case, special diagnostic testing may be advised in addition to newborn screening.
Testing and Follow-Up
The morbidity from congenital hypothyroidism can be reduced to a minimum by early diagnosis and treatment. Although initial preliminary studies were performed using thyroid-stimulating hormone (TSH) levels in cord blood, mass screening was made feasible by the development of radioimmunoassay for TSH and thyroxine (T4) from blood spots on filter paper, obtained for neonatal screening tests.
Screening:
An out-of-range result may occur because the initial blood sample was too small or the test was performed too early. However, as a few babies do have the condition, it is very important that you go to your follow-up appointment for a confirmatory test. Because the harmful effects of untreated CH can occur soon after birth, follow-up testing must be completed as soon as possible to determine whether or not your baby has the condition.
NOTE: THE SERUM TESTING OF CH IS AVAILABLE IN EVERY LAB IN PAKISTAN. The results of Borderline cases of values ranging from 9 to 18 micro-units per mili litres are supposed to be sent for Serum testing to confirm the results before starting of treatment of L-Thyroxine. For more details, email at officecoordinator_zbf@yahoo.com
Diagnosis:
- Diagnosis of primary hypothyroidism is confirmed by demonstrating decreased levels of serum thyroid hormone (total or free T4) and elevated levels of thyroid-stimulating hormone (TSH).
- If maternal antibody–mediated hypothyroidism is suspected, maternal and neonatal anti-thyroid antibodies may confirm the diagnosis. Such antibodies are an uncommon cause of congenital hypothyroidism.
- The combination of low or low-normal serum total T4 levels and a serum TSH within the reference range suggests thyroid-binding globulin (TBG) deficiency. This congenital disorder causes no pathologic consequence, but should be recognized to avoid unnecessary thyroid hormone administration.
Thyroid Scanning:
Thyroid scanning is not required to make or confirm the diagnosis of congenital hypothyroidism, but can provide important information about the etiology. On thyroid scanning (using technetium-99m or iodine-123), the absence of radionuclide uptake suggests sporadic athyreotic hypothyroidism but can also occur when uptake is blocked by excess iodide or thyroid receptor blocking antibodies. If no uptake is found on isotope scanning, thyroid ultrasonography may demonstrate thyroid tissue
Thyroid scans can also demonstrate the presence of an ectopic thyroid, such as a lingual or sublingual gland, which is also sporadic. The presence of a bilobed thyroid in the appropriate position or a goiter would suggest either an inborn error of thyroid hormone production or transient hypothyroidism or hyperthyrotropinemia.
Treatment Management
The mainstay in the treatment of congenital hypothyroidism is early diagnosis and thyroid hormone replacement. Optimal care may include diagnosis before age 10-13 days and normalization of thyroid hormone blood levels by age 3 weeks
Supplementation:
Your baby may need to take L-thyroxine pills, a synthetic thyroid hormone, to replace the natural thyroid hormone that his or her body does not make enough of. An endocrinologist can help determine the appropriate amount of L-thyroxine for your baby and write a prescription.
Only levothyroxine is recommended for treatment. Parents should be provided the hormone in pill form and taught proper administration, as follows:
- The pills can be crushed in a spoon; dissolved with a small amount of breast milk, water, or other liquid immediately before administration; and administered to the child with a syringe or dropper
- The pills should not be mixed in a full bottle of formula
- Finally, prolonged heat exposure may reduce the efficacy of l-thyroxine tablets.
Toddlers typically chew the tablets without problems or complaints. Initial dosages of 10-15 mcg/kg/d, equivalent to a starting dose of 50 mcg in many newborns, have been recommended. Equally good developmental results, but with higher thyroid-stimulating hormone (TSH) levels, have been reported with half this starting dose (25 mcg/d).
Some nutritional supplements or drugs are known to interfere with absorption of l-thyroxine. These include:
- Soy protein formulas
- concentrated iron
- calcium, aluminum hydroxide
- Cholestyramine and other resins
- fiber supplements
- Sucralfate
Recommended Follow Up
Clinical evaluation should be performed every few months during the first three years of life along with frequent measurements of serum T4 or free T4 and TSH. The American Academy of Pediatrics recommends the following monitoring schedule [86].
- At two and four weeks after the initiation of l-thyroxine treatment
- Every 1-2 months during the first 6 months of life
- Every 3-4 months between 6 months and three years of age
- Every 6-12 months thereafter until growth is complete
- Four weeks after any change in dose
More frequently if results are abnormal or non-compliance is suspected.
Dietary limitations
If your baby is taking thyroid hormone pills, you may need to restrict the amount of soy and iron in his or her diet. Soy and iron affect how the body absorbs thyroid hormones from the pills. Tell your baby’s doctor if your baby eats soy-based formula or takes iron supplements. Separate the time you administer your baby’s thyroid medication by at least one hour from the time you feed soy formula or iron medication.
Monitoring
Your child will need regular visits to the doctor to check his or her weight, height, development and overall health. Your child will also likely need regular blood tests to check the level of thyroid hormone. Blood tests are usually done every one to three months until age one, and then every two to four months until age three. They can usually be done less often after age three.
Expected Outcomes
- If treatment starts soon after birth, children with primary congenital hypothyroidism (CH) can have healthy growth and development. This is why newborn screening for CH is so important.
- Treatment will need to continue throughout your child’s life. This may require regular check-ups to monitor thyroid hormone levels, growth, and development.
- Some children may still have trouble in school or delayed growth, even with treatment. Children who struggle in school may need extra help or an early intervention program to prepare them for school.
- Delayed treatment can put children at risk for intellectual disabilities, learning disabilities, developmental delays, and delayed growth.
References
- Léger, J., Olivieri, A., Donaldson, M., Torresani, T., Krude, H., Van Vliet, G., … & Butler, G. (2014). European Society for Paediatric Endocrinology consensus guidelines on screening, diagnosis, and management of congenital hypothyroidism. The Journal of Clinical Endocrinology & Metabolism, 99(2), 363-384.
- Hinton, C. F., Harris, K. B., Borgfeld, L., Drummond-Borg, M., Eaton, R., Lorey, F., … & Pass, K. A. (2010). Trends in incidence rates of congenital hypothyroidism related to select demographic factors: data from the United States, California, Massachusetts, New York, and Texas. Pediatrics, 125(Supplement 2), S37-S47.
- Olney, R. S., Grosse, S. D., & Vogt, R. F. (2010). Prevalence of congenital hypothyroidism—current trends and future directions: workshop summary. Pediatrics, 125(Supplement 2), S31-S36.
- Noreen, R., Memon, M. H., Murtaza, G., & Hanif, S. (2016). Frequency of Congenital Hypothyroidism (CH) in Neonates of a Tertiary Care Hospital of Karachi, Pakistan. Annals of Abbasi Shaheed Hospital & Karachi Medical & Dental College, 21(2).
- Mengreli, C., Kanaka-Gantenbein, C., Girginoudis, P., Magiakou, M. A., Christakopoulou, I., Giannoulia-Karantana, A., … & Dacou-Voutetakis, C. (2010). Screening for congenital hypothyroidism: the significance of threshold limit in false-negative results. The Journal of Clinical Endocrinology & Metabolism, 95(9), 4283-4290.
- Ford, G., & LaFranchi, S. H. (2014). Screening for congenital hypothyroidism: a worldwide view of strategies. Best Practice & Research Clinical Endocrinology & Metabolism, 28(2), 175-187.
- Chen, C. Y., Lee, K. T., Lee, C. T. C., Lai, W. T., & Huang, Y. B. (2013). Epidemiology and clinical characteristics of congenital hypothyroidism in an Asian population: a nationwide population-based study. Journal of epidemiology, 23(2), 85-94.
- Raza, H., Riaz, S., Jamal, M., Shirazi, H., & Gul, S. (2013). Congenital Hypothyroidism Newborn Screening-The PIMS Experience. Ann Pak Inst Med Sci, 9(3), 198-200.
- Khurram, I. M., Choudhry, K. S., Muhammad, K., & Islam, N. (2003). Clinical presentation of hypothyroidism: a case control analysis. J Ayub Med Coll Abbottabad, 15(1), 45-9.

Leave a Reply